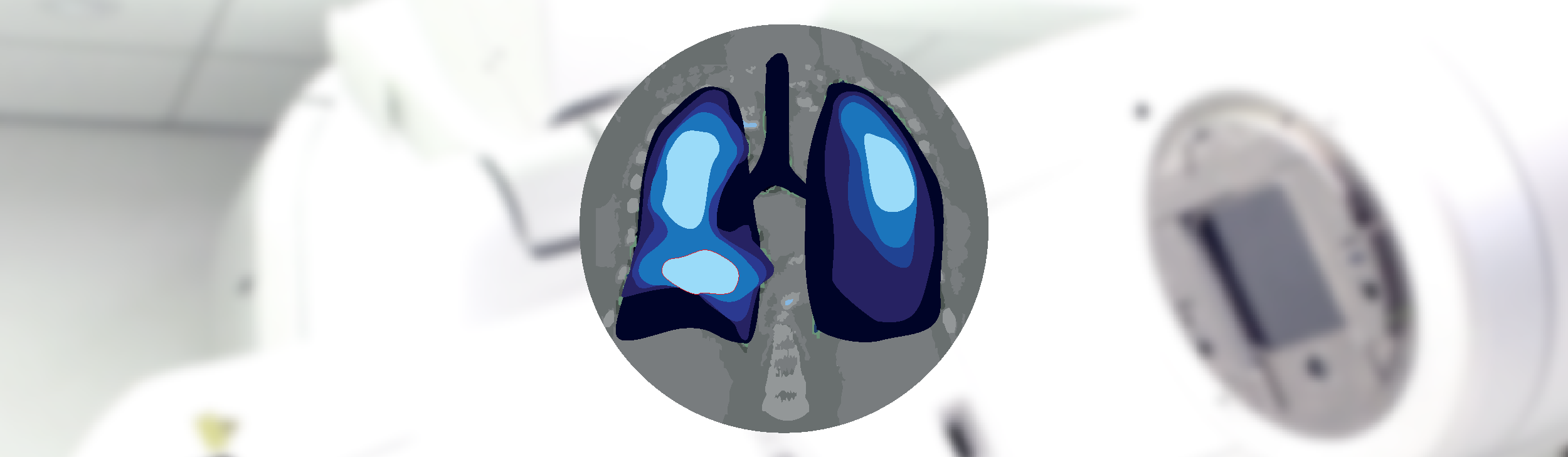
The VITaL Clinical Trial is testing a new method for planning radiation therapy treatments for lung cancer.
IMPORTANT NOTICE: This page is for information only. You cannot enrol in the trial from this page, or through the University of Sydney. You must be the patient of a doctor who is registered for this trial in order to take part. There is a list of participating centres at the bottom of this page.
What are we testing in the VITaL Trial?
The VITaL Trial is testing a new way to protect the healthy parts of your lungs from being damaged by radiation therapy (also known as radiotherapy). The new method is called CT Ventilation Imaging. It maps out which parts of your lungs are the healthiest using your CT scans.
We’ll use this map to carefully guide your radiation therapy treatment, helping to limit any radiation going to the healthy areas of your lungs. Our research predicts that this will mean fewer lung-related side effects from treatment.
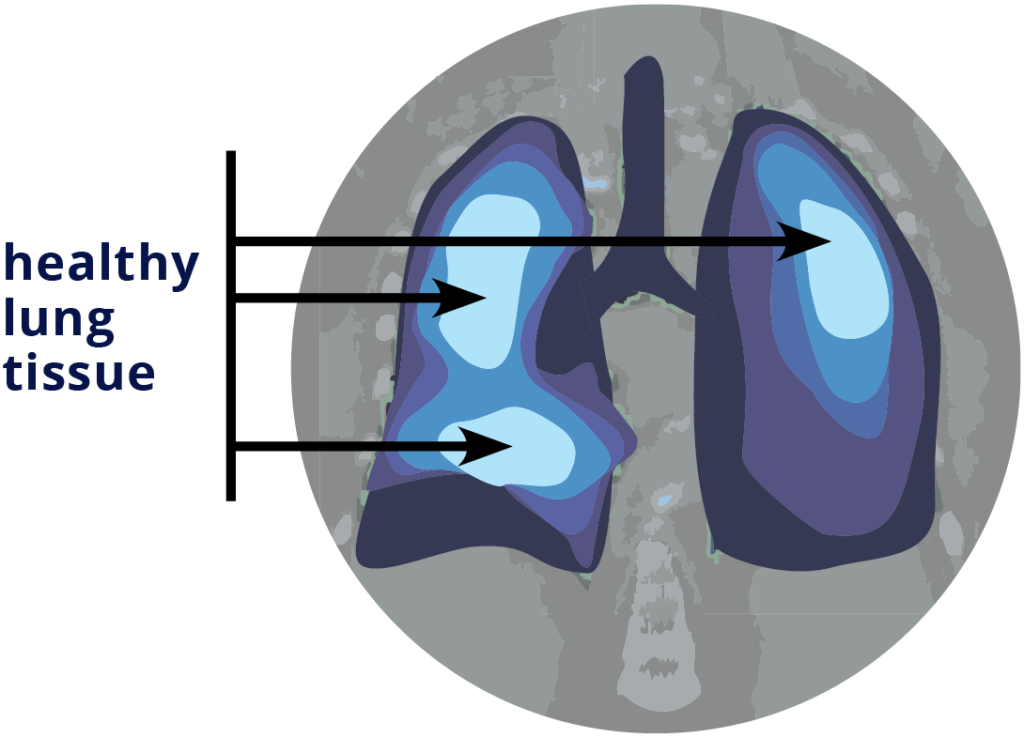
What is the aim of the VITaL trial?
The aim of this trial is to see if our new method of minimising radiation to healthy lung tissue will help to lower lung-related side effects compared to the normal method for lung radiation therapy.
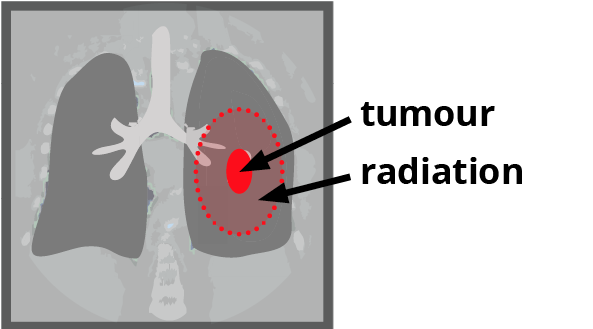
Standard method
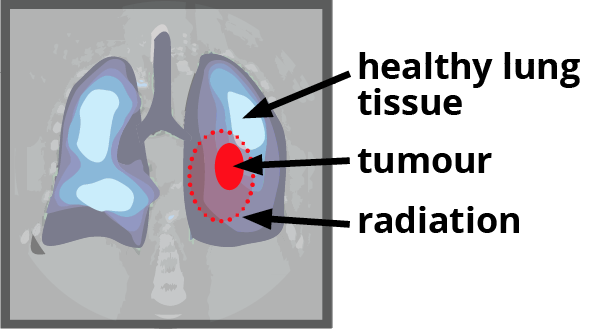
New method
What will taking part mean for me?


How is the trial being run?
Every participant has two treatment plans made from their CT scans – one plan using the standard method, one plan using the new method.
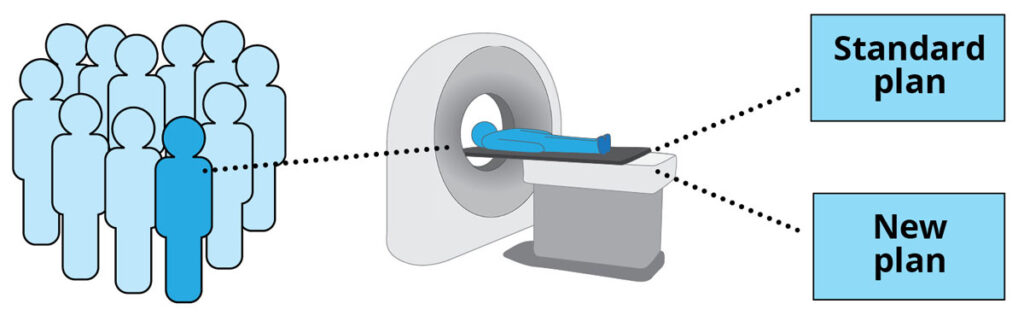
Participants will be put into two groups, randomly assigned by computer – the ‘standard’ group, and the ‘test’ group. The Standard group is treated using the Standard plan. The ‘test’ group is treated using the new plan.
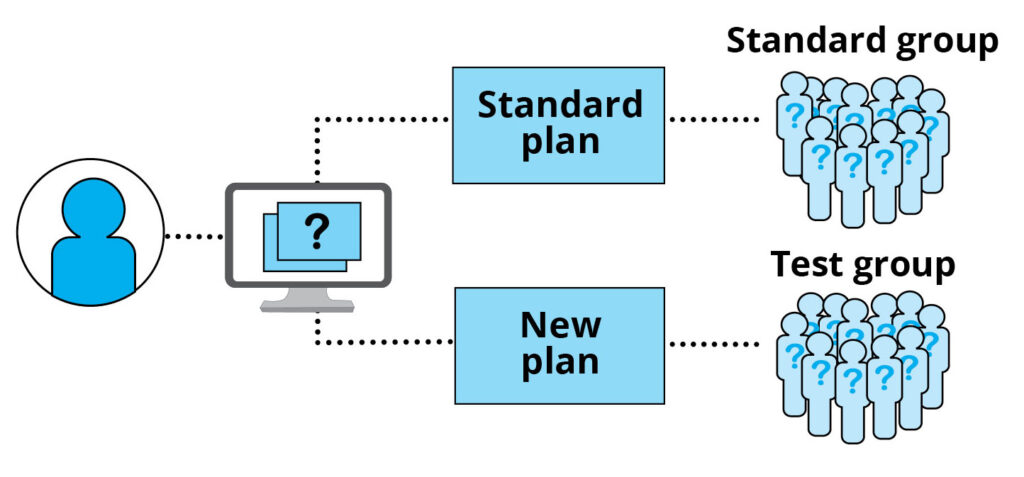
You have a 50/50 chance of being in either group. To keep the results fair and correct, you and your doctor will not know which group you are in. Your doctor can find out which group you are in if it becomes medically necessary.
Am I Eligible to take part?
You may be able to take part if you meet these requirements:
- Aged 18 years or older
- You have recently been diagnosed with, or are suspected to have, Stage 3 non-small cell lung cancer
- You are going to start having radiation therapy for your non-small cell lung cancer
You cannot take part in this research if you are pregnant, trying to get pregnant, or breast-feeding. There is more information about eligibility in the Patient Information and Consent Form.
Taking part in the trial is completely voluntary.
You can pull out at any time, and you don’t have to give a reason why.
Costs and Payments
There are no costs involved in participating – all the scans, tests and care that are part of the research will be provided free of charge.
You will not be paid for taking part in this trial.
What are the risks in taking part?
Radiation therapy has a risk of side effects. Your doctor can talk to you about these. Your overall risk of having side effects from radiation therapy stay the same if you take part in the trial.
If you are in the test group, we expect a reduction in radiation therapy side effects to your lungs. The risk of side effects in other parts of the body will be the same for both the standard and the test groups.
What are the possible benefits of taking part?
If you take part, your treatment plan might avoid giving radiation to the healthiest parts of your lungs. This could mean you’ll experience less side effects.
By joining the VITaL study, you will be part of research that might help other people with lung conditions in the future.
What will happen with the results?
We will present and publish the results of this study in peer-reviewed medical journals and at conferences. Sharing results this way is how research progresses and contributes to the global medical research community. It also provides the pathway for research and new methods to be able to be adopted in healthcare.
Any published materials which display scans or participant data will be de-identified.
We can also share the results of the study with you, if you choose to receive them.
Who is running the trial?

The University of Sydney is the sponsor of the trial. The University of Sydney is responsible for the overall conduct of the study, insurance and indemnity arrangements, and owns the study protocol and study data.
The VITaL Trial is lead by Chief Investigator Professor Paul Keall.
For more information
Your referral doctor or radiation oncologist can discuss the VITaL Trial with you and answer all of your questions.
You can also find detailed information in the Participant Information Sheet and Consent Form – linked below. Please note that the version available on this website may differ slightly from the one used at your participating health care provider.
Contact
Trial Coordinator
Dr Marcel Schulz – [email protected]
Participating Health Care Providers
- Liverpool and Macarthur Cancer Therapy Centres, Liverpool Hospital, Liverpool NSW
- Northern Sydney Cancer Centre, Royal North Shore Hospital, St Leonards NSW
- The Alfred Hospital, Melbourne VIC
- Peter MacCallum Cancer Centre, Melbourne VIC
- GenesisCare Pty Ltd
- Gosford Hospital, Gosford NSW
- Calvary Mater Newcastle NSW
- Wyong Hospital, Wyong NSW
- Canberra Hospital, ACT
- ICON Network
- Sir Charles Gairdner Hospital, WA
- CAPRI Network, Canada
- Sheffield University, UK
This trial has been registered on ClinicalTrials.gov: NCT06127654 and ANZCTR.org.au
This study will be conducted in compliance with all stipulations of this protocol, the conditions of ethics committee approval, the NHMRC National Statement on Ethical Conduct in Human Research (2007) and the Integrated Addendum to ICH E6(R1): Guideline for Good Clinical Practice ICH E6(R2) annotated with TGA comments, effective 09 Nov 2016.
Patient information sheet and Consent Form
Click the link below to download a PDF copy of the Patient information sheet and Consent Form.
Please note that the version available on this website may differ slightly from the one used at your participating health care provider.